 
License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike Located at: (Smith)/Chapter_18:_Electrophilic_Aromatic_Substitution/18.4:_Nitration_and_Sulfonation. Located at: (McMurry)/Chapter_16:_Chemistry_of_Benzene_-_Electrophilic_Aromatic_Substitution/16.01_Electrophilic_Aromatic_Substitution_Reactions:_Bromination. Provided by: Athabasca University Sonoma State University Michigan State U. Steven Farmer Catherine Nguyen Wiliam Reusch, Professor Emeritus. 16.1: Electrophilic Aromatic Substitution Reactions - Bromination.Located at: (Organic_Chemistry)/Arenes/Synthesis_of_Arenes/Electrophilic_Aromatic_Substitution. Synthesis of Benzene Derivatives: Electrophilic Aromatic Substitution.Located at: (Smith)/Chapter_18:_Electrophilic_Aromatic_Substitution/18.2:_The_General_Mechanism. Provided by: Sonoma State University, Michigan State U. Steven Farmer, William Reusch, Professor Emeritus. 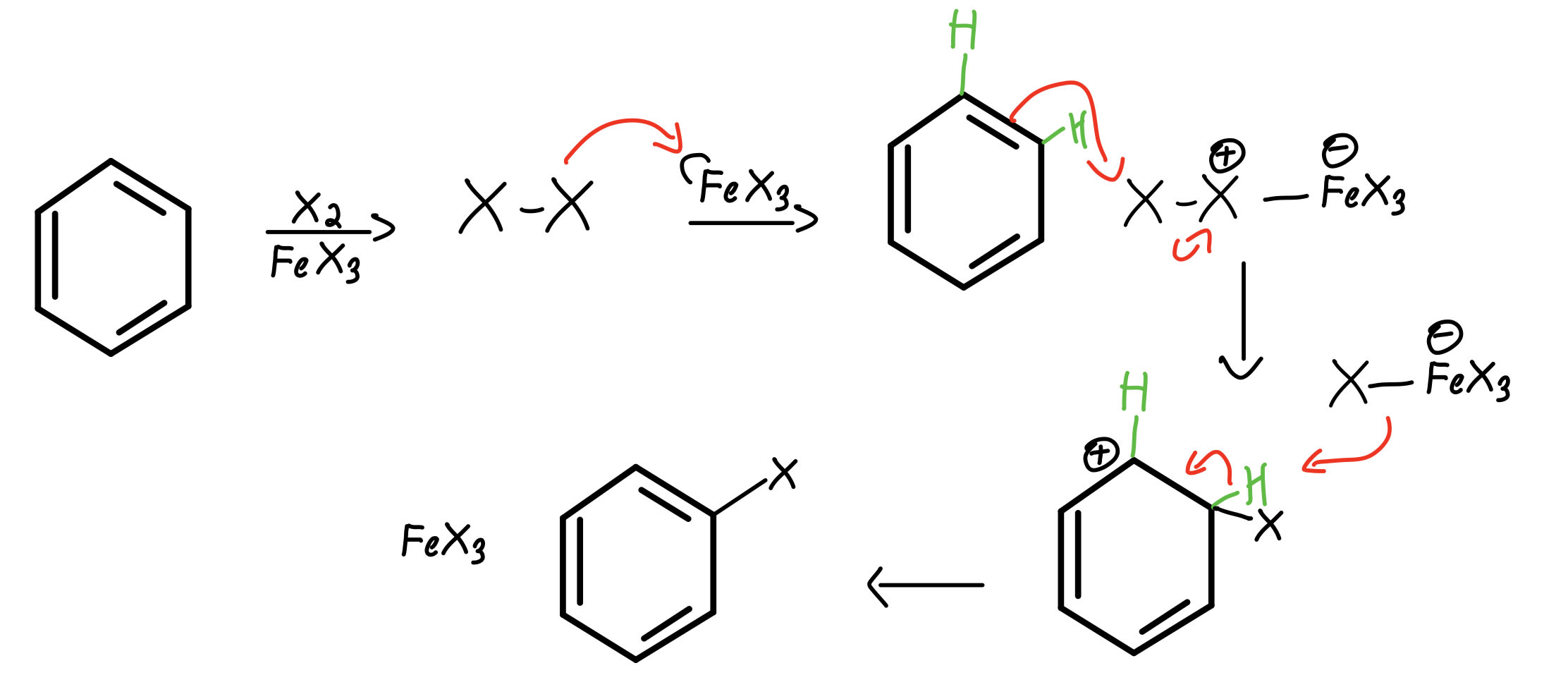
The specific electrophile (E or E ) believed to function in each type of reaction is listed in the right hand column. The catalysts and co-reagents serve to generate the strong electrophilic species needed to effect the initial step of the substitution. Common reactions that proceed by electrophilic aromatic substitution include the nitration and sulfonation of benzene, hydration of benzene, Friedel-Crafts acylation and Friedel-Crafts alkylation. This section is on the general mechanism of how an electrophilic atom becomes a part of a benzene ring through the substitution of a hydrogen. In principle it could react by either mode 1 or 2, but the energetic advantage of reforming an aromatic ring leads to exclusive reaction by mode 2 ( i.e., proton loss). The carbocation intermediate in electrophilic aromatic substitution (the Wheland intermediate) is stabilized by charge delocalization (resonance) so it is not subject to rearrangement. The second step of alkene addition reactions proceeds by the first mode, and any of these three reactions may exhibit molecular rearrangement if an initial unstable carbocation is formed. S N1 and E1 reactions are respective examples of the first two modes of reaction. The cation may rearrange to a more stable carbocation, and then react by mode #1 or #2. The cation may transfer a proton to a base, giving a double bond product (electrophile elimination).ģ. The cation may bond to a nucleophile to give a substitution or addition product (coordination).Ģ. To summarize, when carbocation intermediates are formed one can expect them to react further by one or more of the following modes:ġ. These include S N1 and E1 reactions of alkyl halides, and Brønsted acid addition reactions of alkenes. This mechanism for electrophilic aromatic substitution should be considered in context with other mechanisms involving carbocation intermediates. Further applications of nitration and sulfonation.Sulfuric acid activation of nitric acid.Electrophilic aromatic substitution reactions – Halogenation.Synthesis of benzene derivatives via electrophilic aromatic substitution.  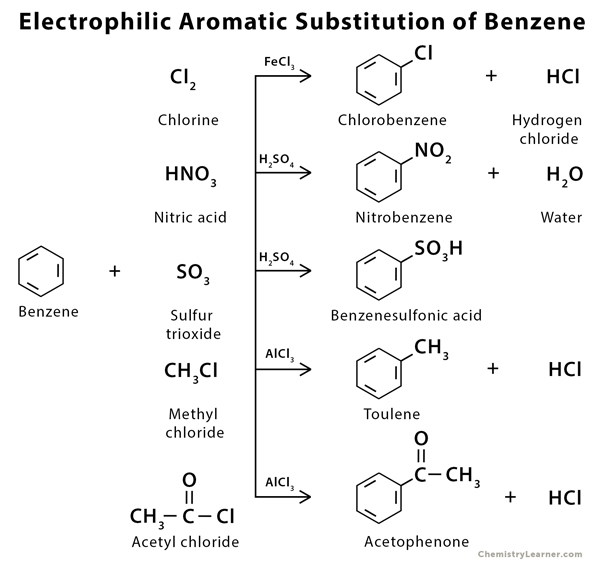
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
